 |
Heat
Transport in Melts, Minerals, Glasses, and
Rocks: Implications for Magma Genesis |
Anne M. Hofmeister1, Alan G. Whittington2 & Peter
I. Nabelek2
1Department
of Earth and Planetary Sciences, Washington University,
St. Louis, MO, 63130 USA, hofmeist@levee.wustl.edu
2Department
of Geological Sciences, University of Missouri, Columbia,
MO, USA, whittingtona@missouri.edu ; nabelekp@missouri.edu
This webpage is
a synopsis of the paper: Whittington,
A.G., A.M. Hofmeister & P.I. Nabelek, Temperature-dependent
thermal diffusivity of the Earth’s crust and
implications for magmatism, Nature, 458,
319-321, 2009, doi:10.1038/nature07818.
For News & Views comment, click here.
Summary
Magmatic processes are dynamic,
so understanding and modeling these complex events
requires data on the thermal transport properties
of not only the surrounding terrain, but also of
the melts themselves. Recent
advances in laser-flash analysis permit accurate measurement
of thermal diffusivity (D) of Earth materials
as a function of temperature (T). They show
that D decreases more strongly with T than
previously thought. Moreover, D decreases,
sometimes drastically, from crystal to glass to melt
of the same composition. Both aspects of thermal
diffusivity are conducive to melting. For example,
our recent thermal model of continental collisions
indicates that D depending on T enhances the
impacts of strain heating or of radiogenic emissions
in producing deep crustal melts.
Introduction
The response of a material
to applied heat depends on its physical properties,
such as the melting temperature. How fast heat travels
through any given material is particularly important
because materials that transport heat slowly stay
warm for longer periods. Thermal
history is governed by the physical property thermal
conductivity (k) through Fourier’s equations.
Earth materials are generally opaque
in appearance. However, our eyes sense the visible,
rather than the near-infrared spectral region, which
is the energy region associated with geologic temperatures. Rocks,
minerals and their glasses and melts are transparent
in the near-IR, even if colored, e.g., by
Fe ions. Because
of these spectral characteristics, laboratory measurements
of heat flow involve some portion of the applied heat
being carried from the heater across the material to
the sensor by light (photons) and some portion being
moved from atom-to-atom within the solid via vibrations
(phonons; Figure 1). Photon transfer in the
laboratory is not a diffusional process, as a material
need not even be present. In most experiments,
no means exists to differentiate whether the sensor
(generally a thermocouple) is heated by phonons diffusing
or photons hopping across the sample (see summary in
Hofmeister et al., 2007).
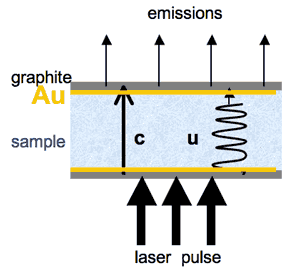
Figure 1: Schematic of heat transfer
in a material that is transparent over some spectral
region. A sample disk (blue stipple
with coated top and bottom) of thickness L is
held at temperature by the furnace. Emissions
from the top of the sample (light arrows) are monitored
by an IR detector, providing a baseline, which is set
to null intensity for convenience (Figure 2). A
laser pulse (heavy arrows) heats the coated bottom
of the sample.
Technology Transfer
Laser-Flash Analysis (LFA, Parker
et al., 1961) is
the industry standard for measuring thermal diffusivity
(D) which is the rate at which cooling occurs
and is closely related to thermal conductivity. Because k =
ρCPD, where ρ is density
and CP is
heat capacity, and these later properties are easily
measured or calculated, LFA measurements provide the
key variable needed to quantify heat transport.
In LFA, heat flow across a sample
is recorded as a function of time (Figure 2). Arrival
times of the photons and phonons are easily distinguished
because of a factor of 100,000 difference in speeds.
The need to constrain heat transport in glasses and
other highly transparent optical materials for applications
in engineering and materials science motivated development
of mathematical models (Mehling
et al., 1998) that
quantitatively remove spurious radiative transfer effects.
Metal and graphite coatings are also used to reduce
photon flux (Degiovanni et al., 1994). These
recent advances permit accurate measurement of thermal
diffusivity for rocks, minerals, glasses and melts.
The technique has additional advantages in studying
geologic materials such as being contact free and quantifying
orientational differences (Hofmeister et al., 2007).
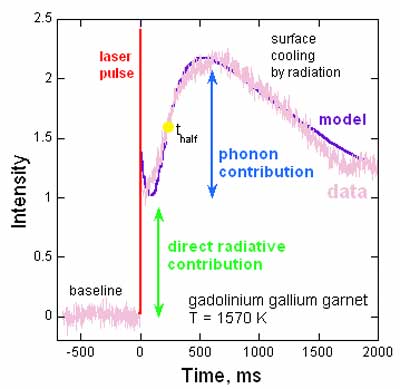
Figure 2. Time-temperature curves in LFA. Measured
emissions = pink. The immediate rise after the
laser pulse (red) is due to direct radiative transfer
(green). Diffusion of heat by vibrations provides a
slower rise (blue) and can be estimated from the half
rise time (yellow dot). Emissions increase to a maximum,
and then the sample radiates heat to the surroundings
until equilibrium is again attained. Purple
= the mathematical model of Mehling et al. (1998) which
accounts for both processes. After Hofmeister
(2006).
Results
Systematic LFA studies have been made
of several common rock-forming mineral families, structural
analogues for high-pressure phases, and various glasses,
melts, and rocks (e.g., Hofmeister, 2006;
Perterman et al., 2008;
Whittington et al., 2009). Our results are
sufficiently accurate to reveal phase transitions and
distinguish between the effects of grain size and porosity
(Branlund & Hofmeister,
2008). We have
found that previous methods underestimated D at
298 K due to contact losses by 10% per thermal contact,
but at high temperature D has
been seriously overestimated, sometimes by a factor
of 5 (Perterman & Hofmeister,
2006; Branlund
& Hofmeister, 2007; Hofmeister
et al., 2007).
Nowhere are the strongly insulating
properties of geologic materials more evident than
in our measurements of glasses and melts (Figure 3).
We have found that isochemical melting reduces the
thermal diffusivity by a non-trivial amount for felsic
minerals and rocks (Perterman
et al., 2008; Whittington
et al., 2009) whereas isochemical melting in mafic
rocks drastically reduces D (Hofmeister
et al., 2009).
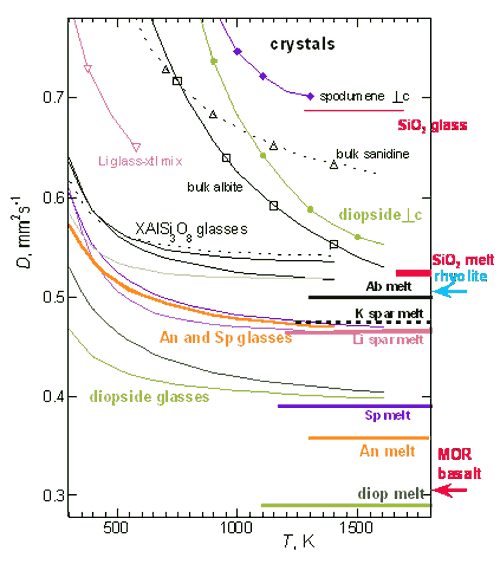
Figure 3. Comparison of thermal
diffusivities. Melts shown as thick lines, glasses
as thin lines, and crystals as very fine lines with
symbols. Compositions are as labeled and the same
type (or color) of line was used for all three states.
Spodumene = purple. Diopside = green. Li-feldspar
= light pink. K-feldspar = black, widely spaced dots.
Albite = black, solid. Anorthite = orange. Remelts
of diopside and albite are shown as darker colors.
Crystals in the Li mixture are spodumene, due to
incongruent melting. Silica = hot pink (Hofmeister,
Branlund and Whittington, in prep.) Rhyolite (Romine,
2008) and basalt (Galenas et al., 2008) studies are
ongoing. Modified from Hofmeister
et al. (2009).
Companion measurements of viscosity
(Hofmeister
et al.,
2009) complete the picture of the transport
properties of melts. We have found that the two properties
are related through the configurational changes (see
discussion of heat capacity, e.g.,
Richet & Bottinger, 1995). Observation
of systematic behavior in the feldspar and pyroxene
systems allows us to estimate D for real magmas
(Figure 3). Current studies of natural basaltic and
rhyolitic lavas (Romine, 2008; Galenas
et al., 2008) support the estimates.
Implications
Because previous data on D (or k)
did not reveal the large difference between
low and high temperature heat transport, constant values
have largely been used in models of heat flow, whether
conduction or convection is involved. Consequently,
the insulating nature of hot rocks has been seriously
underestimated whereas cold rocks have been considered
to retain much more heat than is truly possible (see
discussions by Yuen et al., 2000 and Braun, 2009).
The effect of the variable D on
the thermal evolution of the continental lithosphere
was examined in a thermal model of continental collision
(Whittington et al., 2009) that builds on
previous models based on constant D (e.g.,
Nabelek and Liu, 2004). We found that strain heating
in the shear zones was trapped by the highly insulating
rock, and that crustal melting does not require extraordinary
high levels of radioactivity. The thermal properties
of the rocks and positive feedback led to generation
of leucogranite melts in the Himalayas and other collisional
orogens. Our model does not include the enhanced insulation
provided once melt is formed. For the case studied,
the rocks at high T alone provide enough insulation
to reach melting temperatures, given the presence of
strain heating. For continental hot spots, the
need for a long-term flux of mafic magma to produce
the observed quantities of silicic magma (Annen
& Sparks, 2002; Annen
et al., 2006) should
be reduced when the low thermal diffusivity of hot
crust is taken into account.
Our data on feldspar and pyroxene
crystals, glasses and melts (Figure 3; Hofmeister
et al., 2009) point to mafic systems undergoing
more dramatic changes in thermal properties on melting.
For this case, the difference of rock and melt is key
because at mantle temperatures, D is
more or less independent of temperature. Once partial
melting commences, heat transport by conduction is
impeded. This represents a positive feedback effect,
because heat retention will increase the melt fraction
which in turn further decreases thermal diffusivity
and conductivity of the melting zone. The very large
difference in D between pyroxenes and their
melts (and anorthite and its melt), which proxy for
mafic systems (Figure 3) may contribute to the large
melt fractions that are associated with production
of basaltic magma. Large melt fractions are also more
easily mobilized and extracted, processes that are
also promoted by the low viscosity of basalts. The
different styles of felsic and mafic magmatism are
tied in part to contrasting behavior in both transport
properties (D and viscosity).
What does this mean for plumes?
We
suggest that for the oceanic lithosphere, which is
near solidus everywhere at its base, a small perturbation
in the system will produce magmatism connected with
ridges and hot spots. Huge amounts of heat carried
from the core-mantle boundary are probably not needed
to produce longevity in basaltic magmatism or its huge
volumes, due to thermal feedback provided by exceedingly
insulating melts. Several possibilities come to mind:
the heat could come from the upwellings in an upper
mantle circulation system, or geotherms in the upper
mantle system being slightly uplifted by upwellings
in lower mantle circulation (see patterns in Hofmeister & Criss,
2005). For hot spots such as Iceland, lower solidus
temperatures associated with chemical heterogeneities
in the upper mantle, may provide a nudge. Hydration
alone reduces thermal diffusivity (Hofmeister
et al., 2006). Additional
measurements and thermal models are needed to test
these hypotheses.
References
-
Annen C and Sparks RSJ (2002) Effects of repetitive
emplacement of basaltic intrusions on thermal evolution
and melt generation in the crust. Earth
and Planetary Science Letters, 203,
937-955.
-
Annen C, Blundy JD and
Sparks RSJ (2006) The genesis of intermediate and
silicic magmas in deep crustal hot zones. Journal
of Petrology, 47, 505-539.
-
Branlund J.M. and Hofmeister
A.M. (2007) Thermal diffusivity of quartz to 1000
degrees C: Effects of impurities and the a-b phase
transition. Phys.
Chem. Minerals, 34, 581-595.
-
-
Braun, J. (2009) Hot blanket
in Earth’s
deep crust. Nature, 458, 292-293.
-
Degiovanni A Andre
S Maillet D (1994) Phonic conductivity measurement
of a semi-transparent material. In: Tong TW (ed)
Thermal conductivity 22. Technomic, Lancaster,
PN, pp 623-633.
-
Galenas, M.G., Whittington, A., and Hofmeister,
A., 2008. Transport Properties of Mid-ocean Ridge
Basalt from the East Pacific Rise. IAVCEI 2008 General
Assembly, Reykjavik, Iceland.
-
Hofmeister, A.M. (2006)
Thermal diffusivity of garnets to high temperature. Physics
and Chemistry of Minerals,
33, 45-62.
-
Hofmeister, A.M and Criss, R.E.
(2005) Mantle convection and heat flow in the triaxial
Earth. In: Plates,
Plumes, and Planetary Processes, edited
by G. R. Foulger, J.H. Natland, D.C. Presnall,
and D.L. Anderson, Geological Society of America
Special Paper 388, pp 289-302.
-
Hofmeister A.M., Pertermann,
M., Branlund,
J. and Whittington, A.G. (2006) Geophysical implications
of reduction in thermal conductivity due to hydration. Geophysical
Research Letters, 33, doi:
10.1029/2006GL026036.
-
Hofmeister,
A.M., Pertermann, M. and Branlund, J. M. (2007)
Thermal conductivity of the Earth. Treatise
in Geophysics, G. Schubert, Ed. In Chief,
V. 2 Mineral Physics, G.D. Price, ed. Elsevier,
The Netherlands, pp 543-578.
-
-
Mehling H Hautzinger
G Nilsson O Fricke J Hofmann R Hahn O (1998) Thermal
diffusivity of semitransparent materials determined
by the laser-flash method applying a new mathematical
model. Internatl. J Thermophys. 19,
941-949.
-
Nabelek,
P. I., and Liu, M., 2004, Petrologic and thermal
constraints on the origin of leucogranites in collisional
orogens: Trans. Royal Soc. Edinburgh:
Earth Sciences, 95, 73-85.
-
Parker, J.W., Jenkins,
J.R., Butler, P.C., Abbott, G.I. (1961) Flash method
of determining thermal diffusivity, heat capacity,
and thermal conductivity. J. Appl.
Phys., 32, 1679–1684.
-
-
Pertermann M., Whittington A.G.,
Hofmeister A.M., Spera F.J., and Zayak J. (2008)
Thermal diffusivity of low-sanidine single-crystals,
glasses and melts at high temperatures. Contrib.
Mineralogy and Petrology, 155,
689-702 DOI: 10.1007/s00410-007-0265-x.
-
Richet P and Bottinga Y (1995) Rheology and configurational
entropy of silicate melts. Reviews
in Mineralogy,
32, 67-94.
-
Romine, W.L., 2008. Flow
and heat transfer properties of Mono Craters rhyolites – effects
of temperature, water content and crystallinity.
Unpublished MS thesis, University of Missouri,
121 pp.
-
Whittington AG, Hofmeister AM,
Nabelek PI (2009) Temperature-dependent
thermal diffusivity of Earth's crust: Implications
for crustal anatexis. Nature, 458,
319-321.
-
Yuen,
D.A., Vincent, A.P., Bergeron, S.Y., Dubuffet,
F., Ten, A.A., Steinbach, V.C., Starin, L., 2000.
Crossing of scales and nonlinearities in geophysical
processes. In: Boschi, E., Ekstrom, G., Morelli,
A. (Eds.), Problems in Geophysics
for the New Millenium.
Editrice Compositori, Bologna, Italy, pp. 405-465.
last updated 26th
April, 2009 |
